
Here, we present a review that examines CEA methodologies in depth and discusses how they may affect study findings.Īs of this writing, two literature reviews of the economic value of NSCLC treatment with immunotherapy have been published. 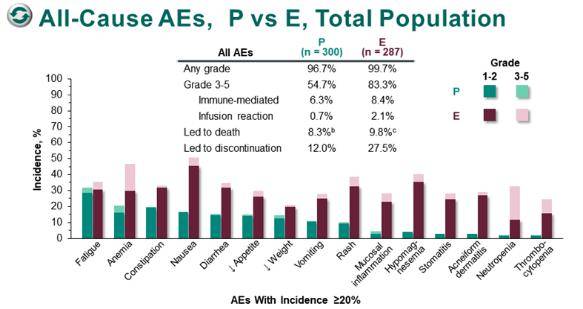
Healthcare payers rely on CEA results to make coverage and reimbursement decisions. 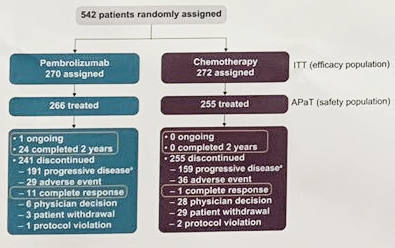
These analyses differed in modeling approaches, survival and cost estimation, and/or utility analyses, yielding varied results and conclusions even for identical patient populations and treatments. The economic value of pembrolizumab-based regimens versus other treatments for NSCLC has been examined in multiple cost-effectiveness analyses (CEAs). Regulatory agencies in many other countries have also approved pembrolizumab with or without chemotherapy as a treatment for all or a portion of these FDA-approved NSCLC indications. First-line pembrolizumab plus chemotherapy combinations are a standard of care for EGFR−/ALK− nonsquamous metastatic NSCLC (KEYNOTE-189) and squamous metastatic NSCLC (KEYNOTE-407). Today, pembrolizumab monotherapy is a first-line standard of care for patients with metastatic NSCLC with a TPS of ≥ 1% and patients with locally advanced NSCLC with a TPS of ≥ 1% who are ineligible for surgical resection or definitive chemoradiation (KEYNOTE-042). Subsequently, first-line pembrolizumab monotherapy was approved for patients with metastatic NSCLC with a TPS of ≥ 50% and no epidermal growth factor receptor or anaplastic large-cell lymphoma kinase ( EGFR−/ALK−) genomic tumor aberrations (KEYNOTE-024). Pembrolizumab, an anti–programmed death-1 (anti–PD-1) monoclonal antibody, was initially approved as a monotherapy by the United States (US) Food and Drug Administration (FDA) in the second- and later-line settings for patients with metastatic NSCLC based on programmed death-ligand 1 (PD-L1) tumor proportion score (TPS) (TPS ≥ 50% based on KEYNOTE-001, and TPS ≥ 1% based on KEYNOTE-010). Immune checkpoint inhibitors are a breakthrough cancer treatment, significantly improving OS and/or progression-free survival (PFS) over chemotherapy for patients with advanced NSCLC in multiple clinical trials. Historically, late-stage NSCLC was treated with chemotherapy, which led to an overall survival (OS) of 8–10 months. Nearly 70% of diagnosed cases are locally advanced or metastatic. NSCLC often does not show symptoms until advanced stages. Non-small cell lung cancer (NSCLC) accounts for 80–85% of all lung cancers and can be further divided into squamous and nonsquamous subtypes. Lung cancer is the most common cancer type and cause of cancer death worldwide. Payers and policy makers should carefully examine study designs and assumptions and choose CEAs with greater validity and accuracy for evidence-based decision-making. Differences in methodology across CEAs may lead to distinct results and conclusions. Consequently, results differed even among studies examining the same population and comparator within similar time intervals. Certain aspects of variability across models were problematic, including deviation from observed treatment utilization within trials and predicted long-term mortality risks for pembrolizumab higher than historical real-world NSCLC mortality data prior to the availability of pembrolizumab. Differences were observed in health states (progression free, progressed disease, and death vs stable disease, progressed disease, death, and treatment discontinuation), modeling approaches (partitioned survival vs Markov), survival extrapolation/transition probability estimation, inclusion of additional costs to drug, disease management and adverse event costs (e.g., programmed death-ligand 1 testing, subsequent treatment, terminal care), treatment duration approaches (trial-based time on treatment vs treat to progression), utility sources (trial data vs literature), and utility analyses (time to death vs progression status). These publications covered regulatory-approved pembrolizumab NSCLC indications based on the following randomized clinical trials: KEYNOTE-010 (one publication), KEYNOTE-024 (six), KEYNOTE-042 (four), KEYNOTE-189 (two), and KEYNOTE-407 (one). Modeling approaches, survival and cost estimation, and utility analyses were compared and evaluated. Fourteen studies were selected through searches of the PubMed database. We review published cost-effectiveness analyses (CEAs) of pembrolizumab as treatment for NSCLC and provide in-depth assessment of their methodologies. 
Pembrolizumab monotherapy or combination therapy is an approved treatment for various advanced non-small cell lung cancer (NSCLC) indications.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
